Isotonix Lawsuit 2026: FDA Warnings, Side Effects, Pyramid Scheme Allegations & Latest Legal Updates
The Isotonix lawsuit (2026 update) has become a major legal and consumer concern involving Market America, the company behind the Isotonix supplement line. Multiple lawsuits, regulatory warnings, and distributor complaints have raised questions about product safety, marketing practices, and income claims.
This updated guide explains the latest lawsuit status, alleged side effects, FDA concerns, pyramid scheme allegations, and settlement developments in a clear and factual format.
What Is Isotonix
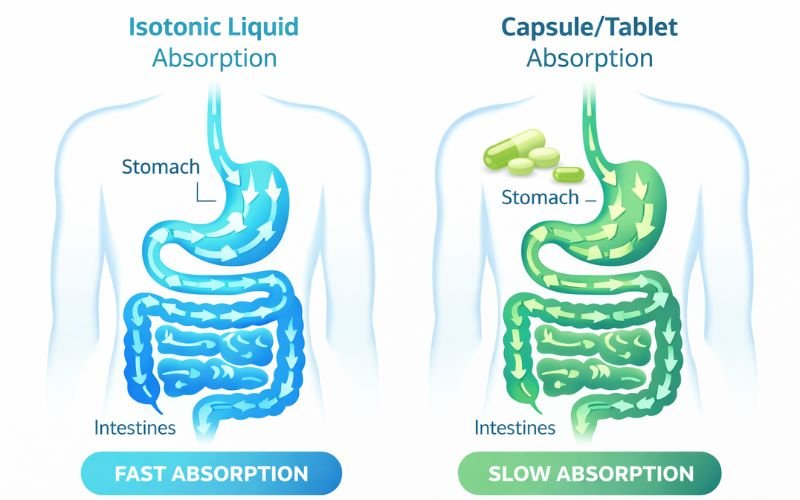
Isotonix is a line of dietary supplements manufactured by Market America and sold through a multi-level marketing (MLM) model.
The products are sold in powdered form and mixed with water. The company claims that its “isotonic delivery system” allows faster absorption of nutrients compared to traditional capsules or tablets.
Popular Isotonix Products Include:
- Isotonix OPC-3 (antioxidant formula)
- Isotonix Multivitamin
- Isotonix Vitamin B12
- Isotonix Activated B-Complex
These products are distributed by independent sellers known as “UnFranchise Business Owners.”
Overview of the Isotonix Lawsuit

The Isotonix lawsuit refers to a series of legal claims filed by consumers and distributors against Market America. These cases focus on allegations that the company misled users about product benefits and business opportunities.
Key Legal Claims Include:
- Alleged false or misleading health claims
- Questionable scientific evidence supporting product benefits
- Lack of proper disclosure of potential risks
- Misleading income representations in the MLM structure
Multiple lawsuits have been filed across different jurisdictions, with some consolidated into larger legal actions.
Key Allegations in the Isotonix Lawsuit
False Health Claims
Plaintiffs allege that Isotonix products were marketed as capable of preventing or treating serious conditions such as heart disease, diabetes, and inflammation. These claims were made without approval from the Food and Drug Administration, which regulates drug-related claims.
Unsubstantiated Scientific Claims
One of the most controversial claims involves the assertion that Isotonix products are absorbed “up to 90% faster” than traditional supplements. Critics argue that there is insufficient scientific evidence to support this claim.
Misleading Income Promises
Former distributors claim they were misled about potential earnings. Many report investing significant amounts in inventory, training, and events, only to earn little or no profit.
Pyramid Scheme Allegations
Some lawsuits argue that the company’s business model relies heavily on recruitment rather than product sales. This raises concerns about whether the structure resembles an illegal pyramid scheme rather than a legitimate MLM business.
FDA and FTC Warnings

In 2020, the Food and Drug Administration issued a warning letter to Market America regarding several violations.
FDA Warning
In prior enforcement actions, the FDA flagged concerns related to:
- Disease-related marketing claims
- Misbranding of supplement products
- Failure to follow reporting requirements for adverse events
FTC Concerns
The Federal Trade Commission has also reviewed MLM marketing practices involving:
- Income exaggeration claims
- Misleading online promotions
- Recruitment-focused messaging
These regulatory concerns have strengthened ongoing legal scrutiny.
Additionally, the Federal Trade Commission raised concerns about misleading online marketing, especially claims related to immunity and disease prevention.
These regulatory actions significantly strengthened legal claims against the company.
Isotonix Side Effects
Common Reported Side Effects
Several lawsuits and consumer reports highlight potential health risks associated with Isotonix products. While not all users experience these effects, the following have been frequently cited:
- Liver problems, including elevated enzyme levels
- Cardiovascular issues such as irregular heartbeat and high blood pressure
- Digestive problems including nausea and stomach pain
- Neurological symptoms like dizziness, headaches, and vertigo
It is important to note that these effects are still under investigation, and no definitive causal relationship has been established.
Isotonix OPC-3 Side Effects
Isotonix OPC-3, a popular antioxidant supplement, has been specifically mentioned in several complaints. Concerns include:
- High antioxidant dosage leading to potential imbalance
- Possible liver strain from concentrated compounds
- Increased risk of headaches and digestive discomfort
Medical experts suggest that excessive intake of certain antioxidants may lead to unintended health complications.
Is Isotonix FDA Approved
No, Isotonix supplements are not FDA-approved.
The FDA does not approve dietary supplements in the same way it approves prescription drugs. Instead, supplement manufacturers are responsible for ensuring product safety and accurate labeling.
However, companies are not allowed to make disease-treatment claims without FDA approval. This is a central issue in the Isotonix lawsuit.
Is Isotonix a Pyramid Scheme
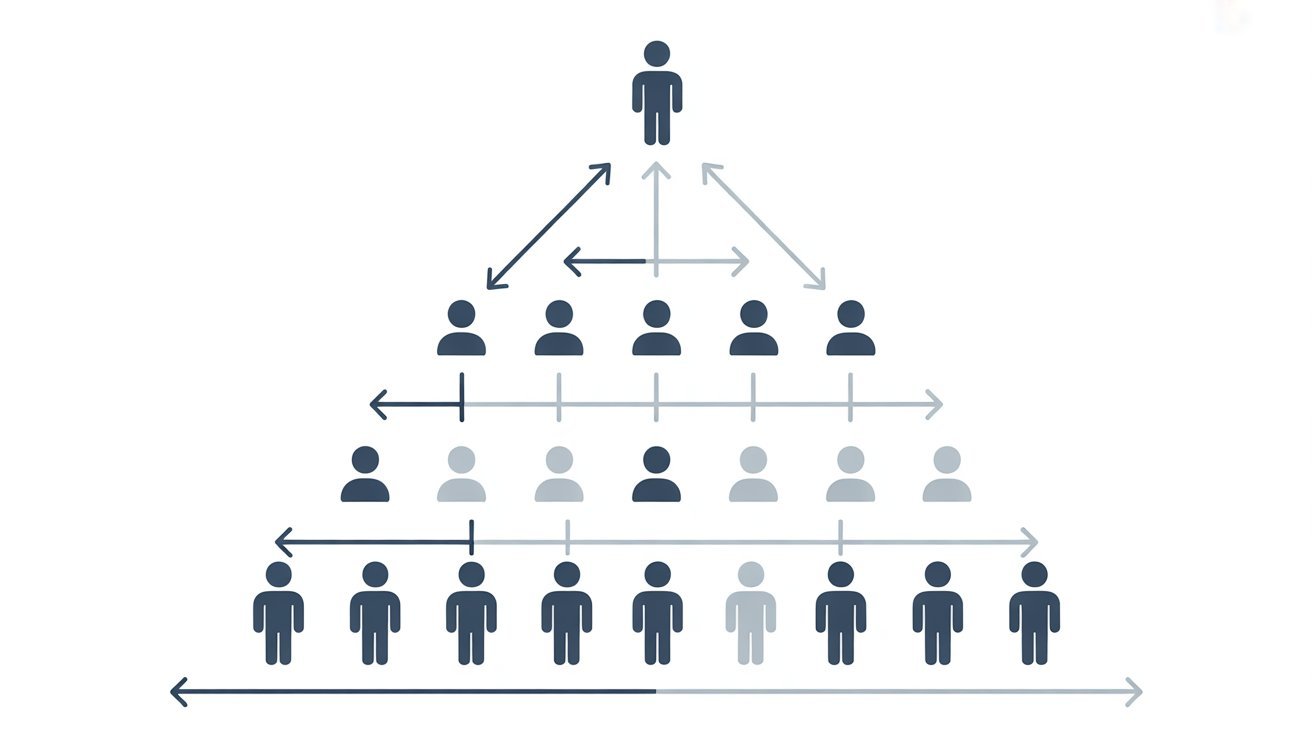
The business model used by Market America operates under a Multi-level marketing structure. While MLMs are legal, they can raise concerns if income depends primarily on recruitment rather than product sales.
MLM vs Pyramid Scheme
- MLM: Revenue is generated from product sales and recruitment
- Pyramid Scheme: Revenue depends mainly on recruitment, with little emphasis on product value
Critics argue that many distributors earn minimal income, while those at higher levels benefit more significantly. These concerns have contributed to ongoing legal scrutiny.
Isotonix Lawsuit Timeline
| Year | Event |
| 2017 | Initial consumer lawsuits filed |
| 2020 | FDA warning issued |
| 2021 | Class action developments |
| 2022 | Company updates labeling and policies |
| 2025–2026 | Ongoing legal proceedings |
Settlement and Latest Updates 2026

As of 2026, the Isotonix lawsuit is still ongoing. There is no confirmed settlement at this time.
Recent developments include:
- Expansion of plaintiff groups
- Continued evidence collection
- Increased regulatory monitoring
Legal experts suggest that settlement discussions may occur, but no official resolution has been announced.
Who Can Join the Isotonix Lawsuit

You may be eligible to participate if:
- You purchased Isotonix products after 2018
- You experienced health issues after use
- You suffered financial losses as a distributor
Steps to Take
- Keep all purchase records and product packaging
- Maintain medical documentation if applicable
- Consult a consumer protection attorney
- Monitor updates on class action eligibility
Compensation Expectations
If the lawsuit results in compensation, affected individuals may be eligible for:
- Medical expense reimbursement
- Lost wages or financial losses
- Compensation for pain and suffering
- Potential punitive damages in severe cases
The final amount depends on the outcome of the case and the severity of individual claims.
Market America’s Response
Market America has denied all allegations related to the Isotonix lawsuit.
The company maintains that:
- Its products are safe and compliant
- Its marketing follows legal guidelines
- Income potential depends on individual effort
In response to regulatory pressure, the company has:
- Updated product labels
- Revised marketing claims
- Improved distributor training
Frequently Asked Questions
What is the Isotonix lawsuit about
The Isotonix lawsuit involves allegations of false advertising, misleading income claims, and potential health risks linked to the supplements.
Is Isotonix safe to use
While many users report no issues, some have reported side effects. It is advisable to consult a healthcare professional before use.
Is Isotonix FDA approved
No, dietary supplements like Isotonix are not FDA-approved.
Can I join the Isotonix lawsuit
You may qualify if you experienced side effects or financial losses related to Isotonix products.
What are OPC-3 side effects
Reported effects include headaches, digestive issues, and possible liver-related concerns.
Conclusion
The Isotonix lawsuit highlights important concerns about supplement safety, marketing transparency, and MLM business practices. While the legal process is still ongoing, the case serves as a reminder for consumers to carefully evaluate health products and business opportunities.
- Supplement marketing practices
- MLM income structures
- Regulatory compliance in the health supplement industry
As more developments emerge in 2026, staying informed will be essential for both consumers and distributors.







